 
All these elements are gases that have no colour and no smell. For example, helium, neon, argon, krypton and xenon are all in the same column and have very similar properties. Elements in the same column, or period, usually have similar properties. This is usually the same as the number of electrons). The periodic table groups elements by their atomic number (how many protons they have. In 1869, Russian scientist Dmitri Mendeleev published the first version of the periodic table. 
In 1905 Albert Einstein used mathematics to prove that the seemingly random movements were caused by the reactions of atoms, and by doing this he conclusively proved the existence of the atom. Brown used Dalton's atomic theory to describe patterns in the way they moved. The pollen grains appeared to be jiggling. In 1827, British scientist Robert Brown looked at pollen grains in water under his microscope. According to Dalton, in a certain compound, the atoms of the compound's elements always combine the same way. He said that compounds are formed when atoms of more than one element combine. Dalton believed that all atoms of the same element have the same mass. In 1803, English philosopher John Dalton suggested that elements were tiny, solid balls made of atoms. Any substance that could be broken down was a compound. He said that an element was any basic substance that could not be broken down into other substances by the methods of chemistry. In 1777 French chemist Antoine Lavoisier defined the term element for the first time. In solid materials, the atoms are tightly packed next to each other so they vibrate, but are not able to move (there is no room) as atoms in liquids do. Because the mass of neutrons and protons is very similar, and the mass of electrons is very small, we can call the amount of protons and neutrons in an atom its atomic mass.Ītoms move faster when they are in their gas form (because they are free to move) than they do in liquid form and solid matter. For example, hydrogen has one proton and sulfur has 16 protons. The number of protons an atom has defines what chemical element it is, this number is sometimes called its atomic number. The nucleus is surrounded by a cloud of light-weight electrons, these are attracted to the protons in the nucleus by the electromagnetic force because they have opposite electric charges. The protons and neutrons are heavier, and stay in the middle of the atom, which is called the nucleus. When atoms join together it is called a chemical reaction.Ītoms are made up of three kinds of smaller particles, called protons, neutrons and electrons. Scientists discover how they work and interact with other atoms through experiments.Ītoms can join together to make molecules: for example, two hydrogen atoms and one oxygen atom combine to make a water molecule. This makes atoms impossible to see without special tools. One nanometer is about 100,000 times smaller than the width of a human hair. 
Atoms range from 0.1 to 0.5 nanometers in width. Examples of elements are hydrogen and gold.Ītoms are very small, but their exact size depends on the element. The chemical elements are organized on the periodic table. These different types of atoms are called chemical elements. 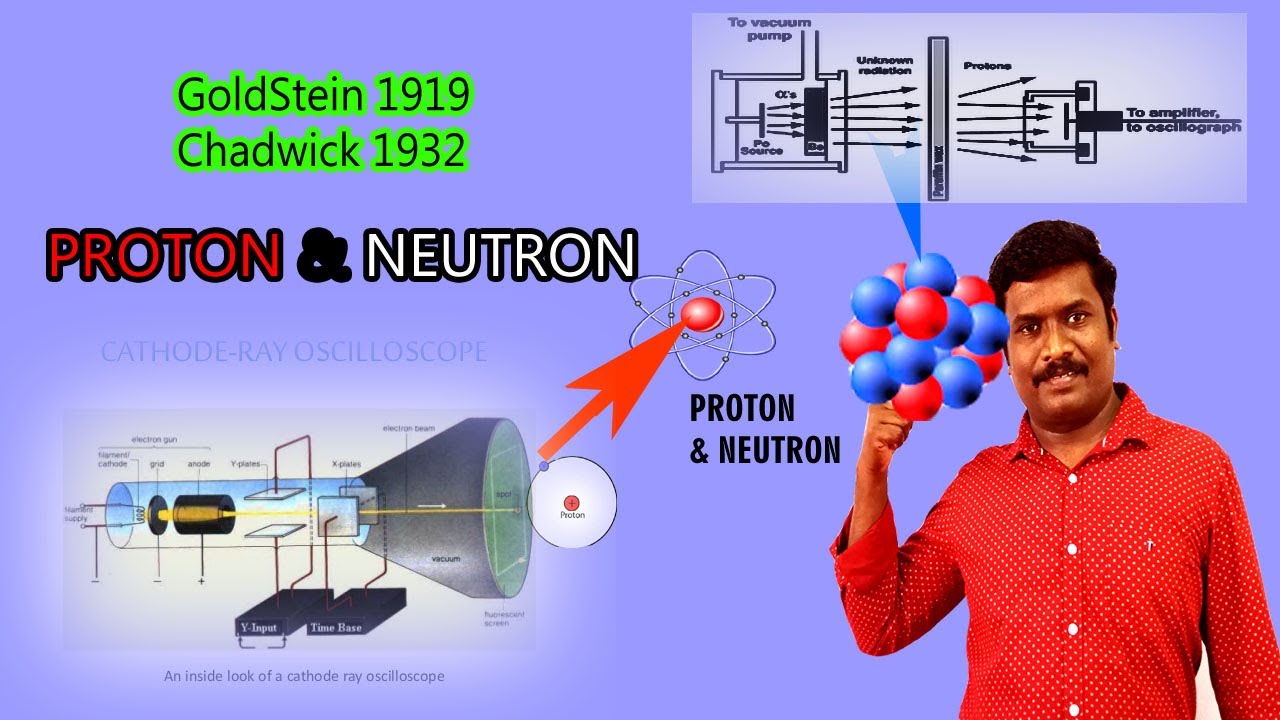
There are many different types of atoms, each with its own name, mass and size. Smallest recognized division of a chemical elementĮlectrons and a compact nucleus of protons and neutronsĪtoms are very small pieces of matter. The black bar is one angstrom ( 10 −10 m or 100 pm). The nucleus (upper right) in helium-4 is in reality spherically symmetric and closely resembles the electron cloud, although for more complicated nuclei this is not always the case. An illustration of the helium atom, depicting the nucleus (pink) and the electron cloud distribution (black). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
